Identification of a protein that inhibits ubiquitination and aggregation of α-synuclein, a causative factor of Parkinson's disease
September 10, 2019
Parkinson's disease is a progressive neurodegenerative disorder characterized by loss of dopaminergic neurons in the substantia nigra and striatum of brains. α-synuclein is the causative protein of Parkinson's disease. Ubiquitinated α-synuclein aggregates in nerve cells play a central role in the development of disease. A research team at Niigata University has discovered that G3BP1 protein inhibits ubiquitination and aggregation of α-synuclein. This study suggested that the G3BP1 plays a protective role in the development of Parkinson's disease by reducing α-synuclein ubiquitination and aggregation. Therefore, G3BP1 is a promising drug target for the treatment of Parkinson's disease.
Publication Details
Title: G3BP1 inhibits ubiquitinated protein aggregations induced by p62 and USP10
Journal: Scientific Reports
Authors: Sergei Anisimov, Masahiko Takahashi, Taichi Kakihana,Yoshinori Katsuragi, Hiroki Kitaura, Lu Zhang, Akiyoshi Kakita, Masahiro Fujii
DOI: 10.1038/s41598-019-46237-1
More News
-

Earliest octopuses were giant top predators in Cretaceous oceans
Research results
-
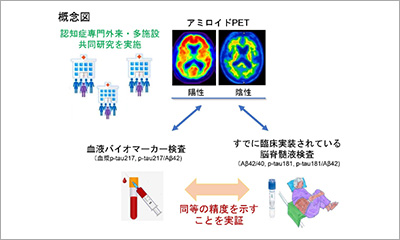
Prospective study on clinical utility of plasma p-Tau217 and other biomarkers in Japanese memory clinics using the LUMIPULSE platform
Research results
-
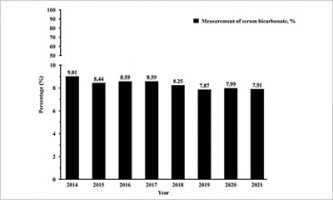
Hidden Acid Imbalance in Kidney Disease Raises Red Flags
Research results
-
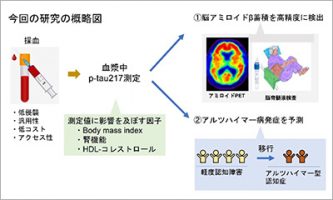
Evaluation of plasma p-tau217 biomarkers in detecting amyloid pathology and predicting cognitive outcomes: Observations from Japanese Alzheimer's disease neuroimaging initiative cohort
Research results
